A. General Theory, Definitions
3. Low Explosives
Low Explosives deflagrate (burn) rather than detonate, they burn progressively, and the reaction is sub-sonic. The low explosives exert more of a pushing or heaving effect (when confined), and are generally used as propellant powders.
Low explosives are sensitive to heat, friction, and shock.
Examples of Low Explosives include: Black Powder, Smokeless Powder, and Flash Powder.
The initiation fuels (A material that yields heat through combustion,) and resultant rapid combustion of the fuel in a confined area. Several fuels when ignited and confined may result in combustion explosion. (Diffuse Fuels)
As with many of the terms associated with explosions, there is also other terms that describe combustion explosions, "Deflagration Explosion". (See Deflagration) The use of either of these descriptors is correct.
a. Fuel Gases
Natural gas, manufactured gas, LP Gas, and similar gases commonly used for commercial or residential purposes, i.e., heating, cooling, or cooking.
commonly used for commercial or residential purposes, i.e., heating, cooling, or cooking.
b. Industrial Gases
A wide variety of products could be included in this list.
c. Dust
It should be noted that a wide variety of materials, even those not generally considered combustible will when in suspension and in the presence of a suitable ignition source will react violently.
d. Smoke
Byproducts of combustion, under the proper conditions can react violently and form a combustion explosion.
6. Detonation
Instantaneous combustion or conversion of a solid, liquid or gas into larger quantities of expanding gases accompanied by heat, shock and a noise.
Detonation is a violent chemical reaction within a chemical compound or mechanical mixture evolving heat and pressure. Detonation is a reaction which proceeds through the reacted material toward the un-reacted material at a supersonic velocity. The result of the chemical reaction is exertion of extremely high pressure on the surrounding medium forming a propagating shock wave that originally is of supersonic velocity. A detonation, when the material is located on or near the surface of the ground, is characterized normally by a crater.
a. Detonation Wave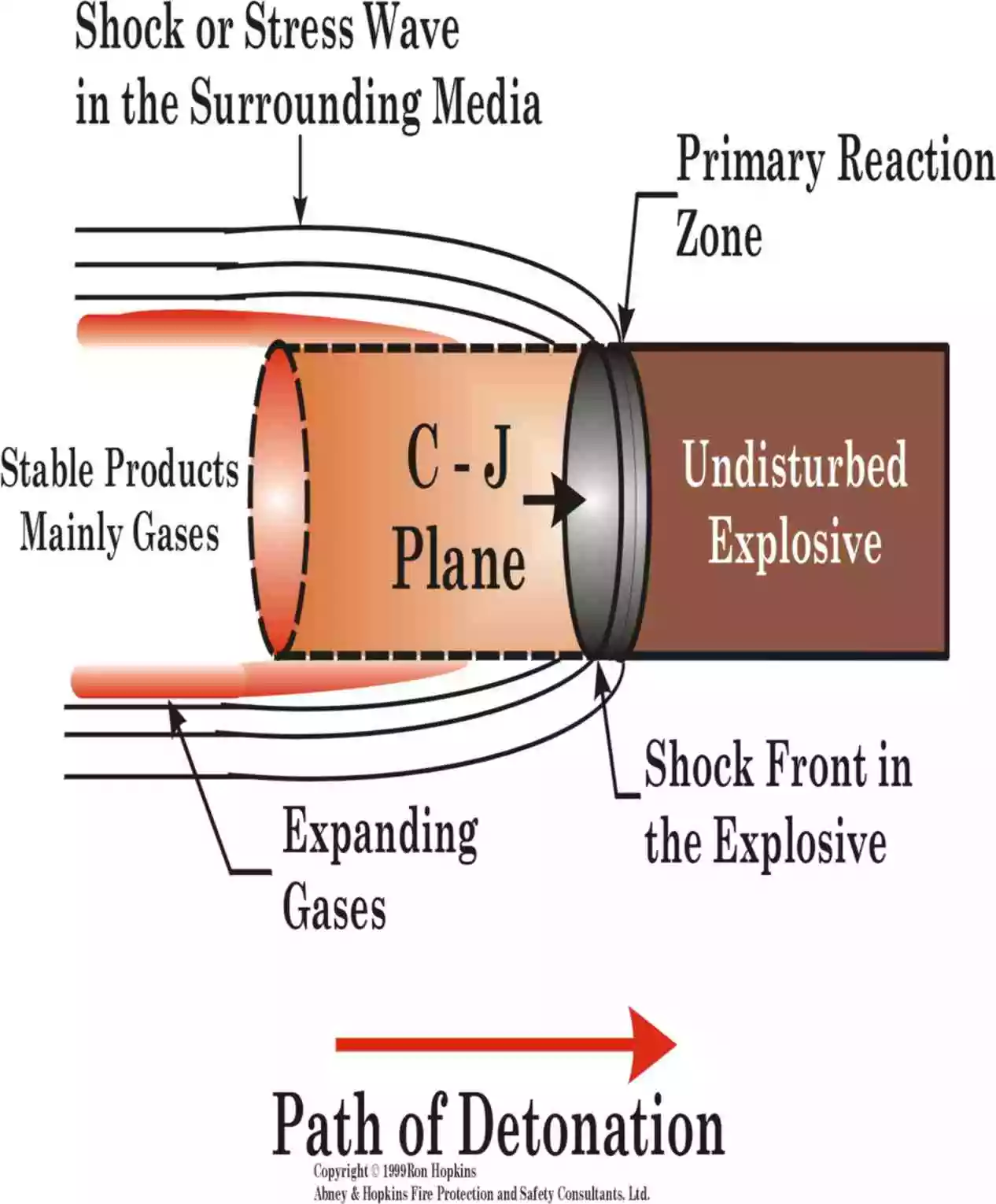
A detonation wave is a shock wave in a reacting (explosive) material where the chemical reaction is carried out in the shock front.
b. Velocity of Detonation
The rate at which the detonation/combustion wave travels through the explosive product. The speed or how fast the chemical reaction occurs or the rate of the reaction. Only High Explosives have a Velocity of Detonation (VOD). The reaction speed is measured in feet/second or meters/second. Velocity of Detonation (VOD)-of a high explosive shock wave will usually vary from 6500 feet/second (2,000 m/second) to 26,000 feet/second (8,000 m/second), the speed of sound being 1085 feet/second (331 m/second) in air. 1 foot = .3048 meters
c. Velocity of Explosion
The rate at which the detonation/combustion wave travels through the explosive product. The speed or how fast the chemical reaction occurs or the rate of the reaction. Velocity of Explosion (VOE) refers to both High and Low Explosives. The reaction speed is measured in feet/second or meters/second
d. Brisance
Brisance is the destructive fragmentation effect of a charge on its immediate vicinity. The relevant parameters in this connection are the detonation rate and loading density (how compact the explosives are) and the heat of explosion and gas yield.
Brisance also describes the shattering effect of an explosive which is a combination of power and velocity. The higher the power and the higher the velocity of detonation, the greater the brisance and shattering effect. High-brisant explosives are used for blasting hard rock and low-brisant explosives are used for blasting where a more pushing or heaving than shattering effect is required.
7. Detonator
In an explosive train, that component which, when detonated by the primer, in turn detonates a less sensitive but larger high explosive (usually the booster); or when containing its own primer initiates the detonation.
A Detonator is any device containing a detonating charge that is used for initiating detonation in an explosive. The term includes, but is not limited to, electric blasting caps of instantaneous and delay types, non-electric blasting caps used with safety fuse, and detonating cord delay connectors.
Blasting cap (Electric or Non-Electric) is often used to denote a detonator.
8. Deflagration
Rapid burning
Deflagration is a rapid chemical reaction in which the output of heat is enough to enable the reaction to proceed and be accelerated without input of heat from another source. Deflagration is a surface phenomenon with the reaction products flowing away from the un-reacted material along the surface at subsonic velocity. The effect of a true deflagration under confinement is an explosion. Confinement of the reaction increases pressure, rate of reaction and temperature, and may cause transition into a detonation.
a. Deflagration to Detonation Transition (DDT)
In detonators where primary explosives are initiated by heat of flame, but are transformed into a detonation.
9. Seated Explosion
The seat of the explosion is defined as the "crater" or "area of greatest damage" that is caused by the explosion. This area is also called the "epicenter" and is the point of origin of the explosion. Initiation of explosives or the area where a mechanical explosion occurs will leave a visible area to indicate the location of the "epicenter". However, often as a result of structural damage, collapse, and similar damage the area of the "epicenter" may be somewhat masked. However, with careful analysis this area can often be detected.
a. Characteristics
Crater
High Pressures
Rapid rates of pressure rise
Detonations - supersonic explosive velocity
b. Fuels or Products Involved
(1.) Explosives
(2.) Steam Boilers
(3.) BLEVE
(4.) Tightly confined Gases or Liquid Vapors
10. Non-Seated Explosion
Those explosions where there is no physical evidence of a single location where the explosion originated. Non-Seated explosions are generally as a result of the ignition of diffuse fuels such as gases (i.e. Natural Gas, LPG, Sewer Gases, Industrial Gases), vapors of pooled liquids (i.e. gasoline vapors, lacquer thinner, MEK), and dusts.
It should be noted, however, that by careful analysis of the scene often there is a definite overpressure direction that can be detected even though there is no crater formation. Also, the epicenter is where the source of ignition is and not the location of the fuel gas leak.
a. Characteristics
No Crater
Wide spread fuels
Moderate rates of pressure rise
Deflagrations, subsonic
b. Fuels
(1.) Fuel Gases
A general category of combustion explosions that occur as a result of the ignition of diffuse fuels such as gases (i.e. Natural Gas, LPG, Sewer Gases, Industrial Gases), vapors of pooled liquids (i.e. gasoline vapors, lacquer thinner, MEK).
(2.) Dusts
Finely divided solid materials such as dusts and fines, when in suspension and ignited, can produce violent and destructive explosions. It should be noted that even materials generally not considered combustible can produce an explosion when burned in suspension.
In that the combustion reaction is a surface reaction, the rates of pressure rise generated by combustion are largely dependent on the surface area of the dispersed dust particles. The finer the dust, the more violent the reaction.
(3.) Pooled Flammable/Combustible Liquids
(4.) Backdraft Smoke

 commonly used for commercial or residential purposes, i.e., heating, cooling, or cooking.
commonly used for commercial or residential purposes, i.e., heating, cooling, or cooking.

